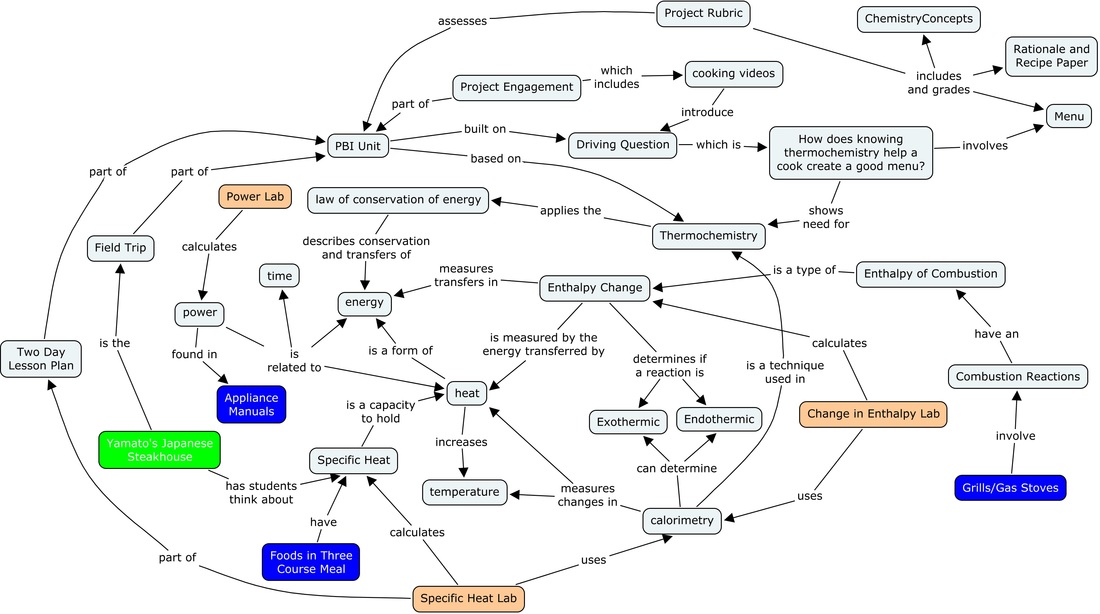
Key: Investigative Lessons are blue, Benchmark Lessons are peach, and the Field Trip is green.
Narrative
Thermochemistry is the study of the transfers of energy as heat that accompany chemical reactions and physical changes. Heat and temperature should never be confused. Temperature is the measure of the average kinetic energy of the particles in a sample of matter, whereas heat is the energy transferred between two samples of matter of different temperatures. A reaction that gives off heat during a reaction, often resulting in an increase of temperature, is termed an exothermic reaction. On the other hand an endothermic reaction absorbs heat, resulting in a decrease in temperature of the system. The energy absorbed or released as heat in a chemical or physical change can be measured in a calorimeter. The amount of energy as heat required to raise one gram of a substance by 1 degree Celsius or Kelvin is defined as that materials specific heat, an intrinsic property of a substance. Heat transfer is often represented by the equation q=mcΔT. If you know any three of the four variables of a reaction (energy transferred in joules,q, the specific heat of a material, c, the amount used, m, and the temperature change) the fourth can be calculated. The energy transferred in a reaction is normally describes in units of joules. It is also important to know the Law of Conservation of Energy, which basically states that energy cannot be created or destroyed, it can only change form.
Enthalpy change is a quantitative way of describing how much energy is released or absorbed in a chemical reaction. The enthalpy values of various reactions can be measured directly through methods such as calorimetry, or indirectly through calculations. The values of the enthalpy, or heat, of combustion are important in for determining heat given off by grills and gas stoves that commonly use propane or methane.
Though power is not usually a chemistry concept, it is crucial for the outcome of this project. Power is the rate at which energy is transferred or transformed, which in the context of cooking appliances is equal to the amount of heat transferred per unit of time. Power is normally described in units of watts (joules/sec). It is important to know the relationship between power and heat since many appliances have power outputs in their specifications.
The focus of this project is on figuring out which cooking appliance is best for cooking the foods for a three course meal. This project uses thermochemistry to come up with a solution to this problem through the concepts of specific heat, enthalpy change, and power.
Enthalpy change is a quantitative way of describing how much energy is released or absorbed in a chemical reaction. The enthalpy values of various reactions can be measured directly through methods such as calorimetry, or indirectly through calculations. The values of the enthalpy, or heat, of combustion are important in for determining heat given off by grills and gas stoves that commonly use propane or methane.
Though power is not usually a chemistry concept, it is crucial for the outcome of this project. Power is the rate at which energy is transferred or transformed, which in the context of cooking appliances is equal to the amount of heat transferred per unit of time. Power is normally described in units of watts (joules/sec). It is important to know the relationship between power and heat since many appliances have power outputs in their specifications.
The focus of this project is on figuring out which cooking appliance is best for cooking the foods for a three course meal. This project uses thermochemistry to come up with a solution to this problem through the concepts of specific heat, enthalpy change, and power.